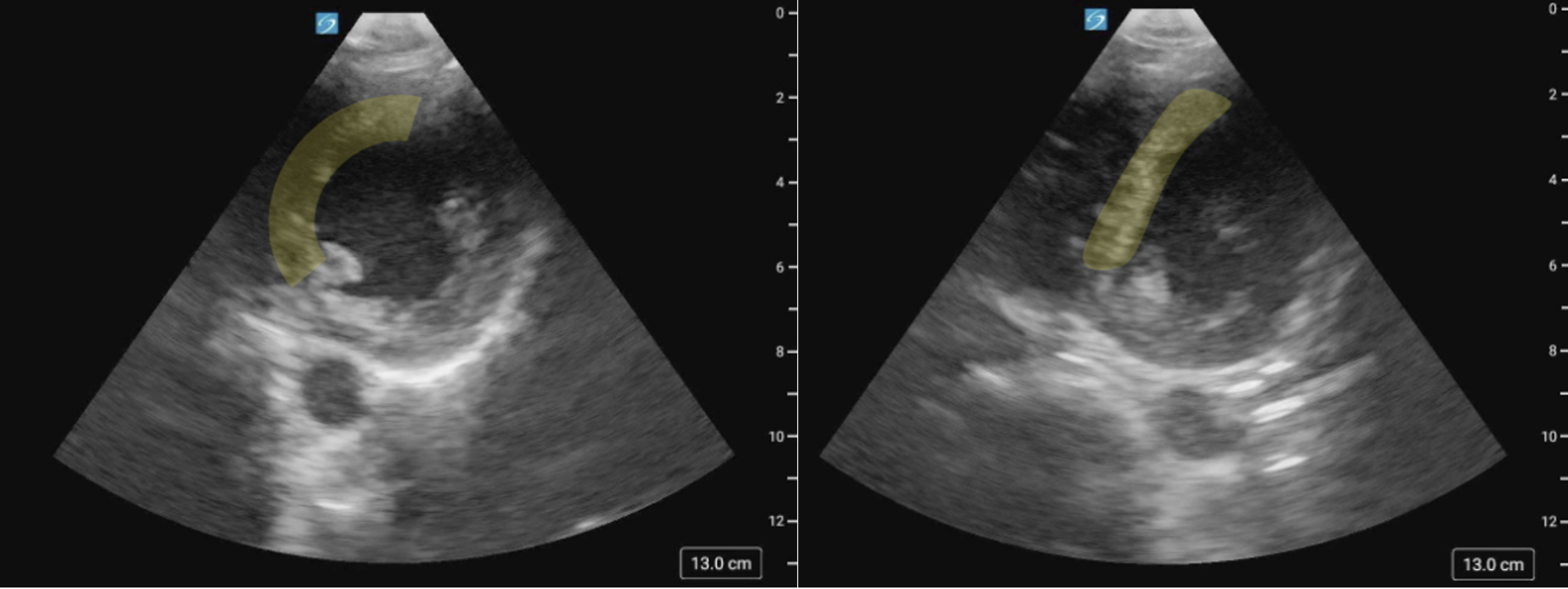
Each cardiac view offers a different perspective of the LV, displaying various walls and myocardial segments. The LV is typically divided into three levels: basal, mid, and apical. The basal segments are located closest to the atrioventricular valves and represent the upper portion of the LV; the mid segments are situated in the middle, at or around the level of the papillary muscles, and the apical segments are near the apex of the ventricle. Understanding which walls are visualized in each view is key to assessing LVfx.
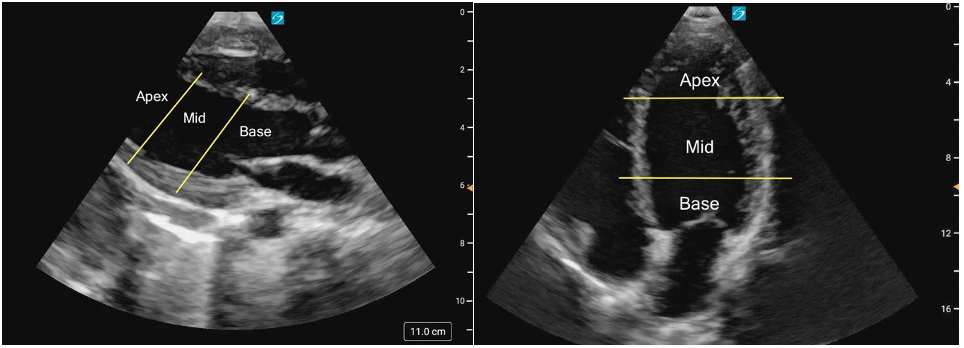 Figure 1ab: LV levels in the PLAX view (a) and A4C (b) in cardiology convention
Figure 1ab: LV levels in the PLAX view (a) and A4C (b) in cardiology convention
At a Glance: Normal VS Not Normal
The findings below outline the broad features of what is considered normal and not normal for pediatric LVfx; the specifics for each individual view will be described in the following sections.
What is Normal
Contractility:
The contraction of the LV occurs in a complex but coordinated manner, combining longitudinal shortening, radial thickening, and circumferential constriction [20]. The base of the heart moves toward the apex as the ventricle shortens, while the cavity simultaneously decreases in diameter due to inward wall motion. This multidirectional contraction pattern efficiently ejects blood into the aorta and gives the ventricle its characteristic torsional motion, where the apex rotates slightly counterclockwise and the base clockwise during systole [20].
In normal systolic function, the LV walls should thicken visibly and move inward symmetrically, coming close to one another during systole. All walls should demonstrate coordinated inward motion and uniform thickening (radial contraction). You should also see the base move toward the apex (longitudinal shortening). Although circumferential shortening contributes to the overall narrowing of the cavity, this is not directly distinguishable on standard PoCUS imaging.
Size and Shape:
The LV should be ellipsoid or bullet-shape. It is typically larger than the right ventricle, assuming the RV is normal, usually about one and a half times the size of the RV.
What is NOT normal
Decreased global function:
LVfx is considered reduced if the LV walls fail to contract effectively toward the center of the cavity during systole. Wall motion will appear sluggish or diminished, and the overall inward movement and thickening will be reduced.
Size and Shape:
Decreased LV systolic function is often accompanied by dilation, such as in dilated cardiomyopathy. The LV loses its normal ellipsoid or bullet shape, appearing globular or rounded, and is much larger than the right ventricle.
RWMAs:
Segmental dysfunction may be observed when one or more LV wall segments move less than expected (hypokinesis), move opposite direction as normal (dyskinetic), or do not move at all (akinesis), while other segments contract normally. These findings often suggest ischemia or myocardial injury. Akinetic segments will also often appear thinner and more hyperechoic than normal myocardium reflecting myocardial fibrosis or scarring that reduce contractility and alter tissue characteristics.
Hyperdynamic function:
In cases of increased contractility, the LV walls may nearly or completely touch during systole, leading to near obliteration of the ventricular cavity. This is often most pronounced at the apex, while the basal segments may not completely obliterate. Hyperdynamic function can occur in states of low preload, hypovolemia, or compensatory hypercontractility. Of note, tachycardia can create the illusion of hyperdynamic function, therefore it is important to look at the wall motion and cavity itself and not just the rate at which the heart is contracting. Saving and slowing down a clip can be helpful in cases of significant tachycardia
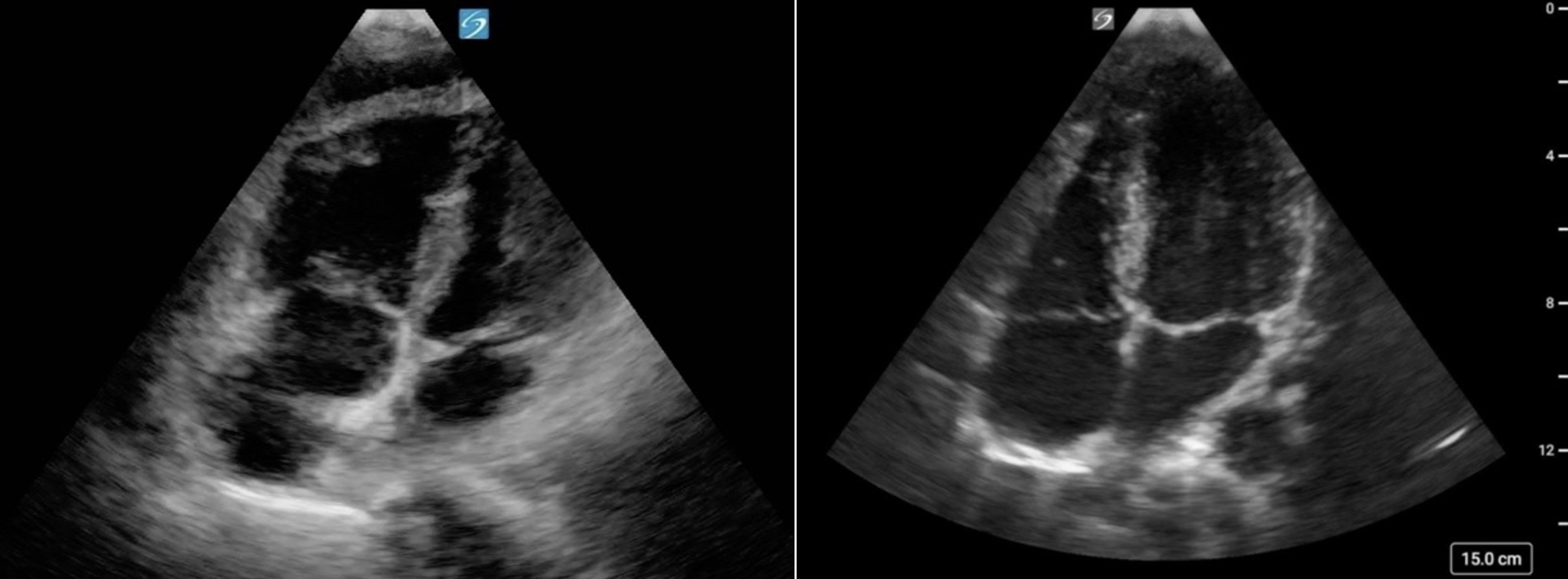
Figure 2a. Normal function seen in the A4C view
Figure 2b. Severely decreased function, globally seen in the A4C view