**To unlock access to the first quiz, make sure to select the “Mark as Completed” button below
References
- Patel, G et al. Point-of-Care Cardiac Ultrasound (POCCUS) in the Pediatric Emergency Department. Clinical Pediatric Emergency Medicine. 2018. 19: 323-327. DOI: 10.1016/j.cpem.2018.12.009
- Guevarra K, Greenstein Y. Ultrasonography in the Critical Care Unit. Curr Cardiol Rep. 2020;22(11):145. doi:10.1007/s11886-020-01393-z
- Volpicelli G, Lamorte A, Tullio M, et al. Point-of-care multiorgan ultrasonography for the evaluation of undifferentiated hypotension in the emergency department. Intensive Care Med. 2013;39(7):1290-1298. doi:10.1007/s00134-013-2919-7
- Potter SK, Griksaitis MJ. The role of point-of-care ultrasound in pediatric acute respiratory distress syndrome: emerging evidence for its use. Ann Transl Med. 2019;7(19):507-507. doi:10.21037/atm.2019.07.76
- Mojoli F, Bouhemad B, Mongodi S, Lichtenstein D. Lung Ultrasound for Critically Ill Patients. Am J Respir Crit Care Med. 2019;199(6):701-714. doi:10.1164/rccm.201802-0236CI
- Longjohn M, Wan J. Point-of-Care Echocardiography by Pediatric Emergency Physicians. Pediatric Emergency Care. 2011;27(8).
- Miller AF, Arichai P, Gravel CA, et al. Use of Cardiac Point-of-Care Ultrasound in the Pediatric Emergency Department. Pediatr Emer Care. 2022;38(1):e300-e305. doi:1097/pec.0000000000002271
- Riera A, Weeks B, Emerson BL, Chen L. Evaluation of a Focused Cardiac Ultrasound Protocol in a Pediatric Emergency Department. Pediatr Emer Care. 2021;37(4):191-198. doi:1097/pec.0000000000001495
- Spurney CF, Sable CA, Berger JT, Martin GR. Use of a hand-carried ultrasound device by critical care physicians for the diagnosis of pericardial effusions, decreased cardiac function, and left ventricular enlargement in pediatric patients. Journal of the American Society of Echocardiography. 2005;18(4):313-319. doi:1016/j.echo.2004.10.016
- Conlon TW, Himebauch AS, Fitzgerald JC, et al. Implementation of a Pediatric Critical Care Focused Bedside Ultrasound Training Program in a Large Academic PICU*. Pediatric Critical Care Medicine. 2015;16(3):219-226. doi:1097/pcc.0000000000000340
- Moore CL, Rose GA, Tayal VS, Sullivan DM, Arrowood JA, Kline JA. Determination of Left Ventricular Function by Emergency Physician Echocardiography of Hypotensive Patients. Academic Emergency Medicine. 2002;9(3):186-193. doi:1197/aemj.9.3.186
- Vorel ES, Jacquemyn X, Cohen JS, Kutty S, Deanehan JK. Pediatric Reference Ranges and Test Characteristics of E-point Septal Separation as a Marker for Left Ventricular Dysfunction: A Retrospective Study. Pediatric Emergency Care. Published online April 14, 2025. doi:1097/pec.0000000000003393
- Griffee MJ, Merkel MJ, Wei KS. The role of echocardiography in hemodynamic assessment of septic shock. Crit Care Clin. 2010;26(2):365-382, table of contents. doi:10.1016/j.ccc.2010.01.001
- Watkins LA, Dial SP, Koenig SJ, Kurepa DN, Mayo PH. The Utility of Point-of-Care Ultrasound in the Pediatric Intensive Care Unit. J Intensive Care Med. Published online October 9, 2021:088506662110478. doi:10.1177/08850666211047824
- Gaspar HA, Morhy SS. The Role of Focused Echocardiography in Pediatric Intensive Care: A Critical Appraisal. BioMed Research International. 2015;2015:1-7. doi:10.1155/2015/596451 de Boode WP, van der Lee R, et al. The role of Neonatologist Performed Echocardiography in the assessment and management of neonatal shock. Pediatr Res. 2018;84(S1):57-67. doi:10.1038/s41390-018-0081-1
- Arnoldi S, Glau CL, Walker SB, et al. Integrating Focused Cardiac Ultrasound Into Pediatric Septic Shock Assessment*. Pediatric Critical Care Medicine. 2021;22(3):262-274. doi:10.1097/PCC.0000000000002658
- Ranjit S, Aram G, Kissoon N, et al. Multimodal Monitoring for Hemodynamic Categorization and Management of Pediatric Septic Shock: A Pilot Observational Study*. Pediatric Critical Care Medicine. 2014;15(1):e17-e26. doi:10.1097/PCC.0b013e3182a5589c
- Singh Y, Tissot C, Fraga MV, et al. International evidence-based guidelines on Point of Care Ultrasound (POCUS) for critically ill neonates and children issued by the POCUS Working Group of the European Society of Paediatric and Neonatal Intensive Care (ESPNIC). Crit Care. 2020;24(1):65. doi:10.1186/s13054-020-2787-9
- Lu JC, Riley A, Conlon T, et al. Recommendations for Cardiac Point-of-Care Ultrasound in Children: A Report from the American Society of Echocardiography. Journal of the American Society of Echocardiography. 2023;36(3):265-277. doi:1016/j.echo.2022.11.010 1.
- Sengupta PP, Tajik AJ, Chandrasekaran K, Khandheria BK. Twist Mechanics of the Left Ventricle. JACC: Cardiovascular Imaging. 2008;1(3):366-376. doi:1016/j.jcmg.2008.02.006
- Engle SJ, DiSessa TG, Perloff JK, et al. Mitral valve E point to ventricular septal separation in infants and children. The American Journal of Cardiology. 1983;52(8):1084-1087. doi:1016/0002-9149(83)90537-4
- Tissot C, Singh Y, Sekarski N. Echocardiographic Evaluation of Ventricular Function—For the Neonatologist and Pediatric Intensivist. Front Pediatr. 2018;6. doi:3389/fped.2018.00079
- Margossian R, Schwartz ML, Prakash A, Wruck L, Colan SD, Atz AM, et al. Comparison of echocardiographic and cardiac magnetic resonance imaging measurements of functional single ventricular volumes, mass, and ejection fraction (from the Pediatric Heart Network Fontan CrossSectional Study). Am J Cardiol (2009) 104(3):419–28. doi:10.1016/j. amjcard.2009.03.058 1.
- Muniz RT, Mesquita ET, Souza Junior CV, Martins WDA. Pulmonary Ultrasound in Patients with Heart Failure – Systematic Review. Arquivos Brasileiros de Cardiologia. Published online 2018. doi:5935/abc.20180097
- van Royen N, Jaffe C, Krumholz H, Johnson K, Lynch P, Natale D, Atkinson P, Deman P, Wackers F. Comparison and Reproducibility of Visual Echocardiographic and Quantitative Radionuclide Left Ventricular Ejection Fractions. American Journal of Cardiology. 1996;77.

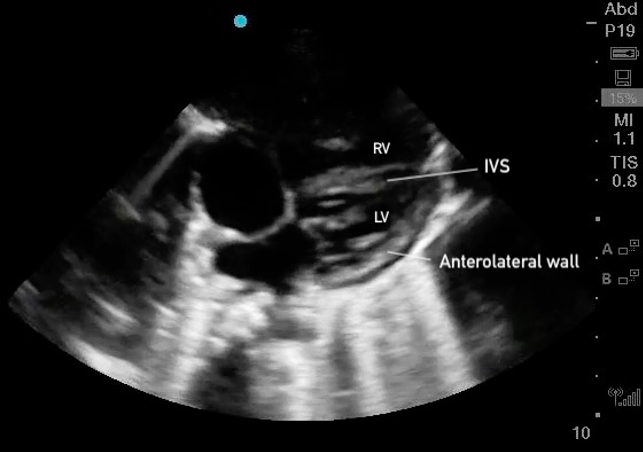 Figure 25. Subxiphoid 4 chamber with the LV walls labeled
Figure 25. Subxiphoid 4 chamber with the LV walls labeled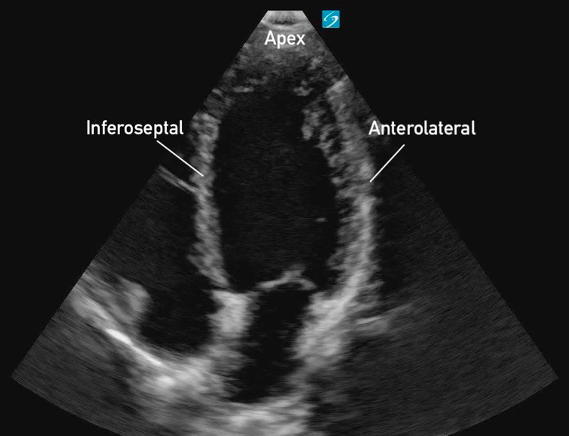 Figure 19: Apical 4 Chamber view in cardiology convention with the labeled left ventricular walls
Figure 19: Apical 4 Chamber view in cardiology convention with the labeled left ventricular walls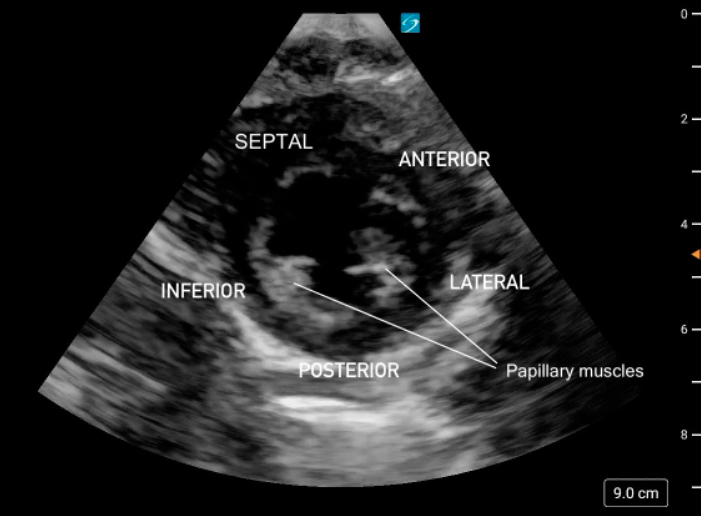 Figure 11. PSAX at the mid-papillary level with left ventricular walls labeled.
Figure 11. PSAX at the mid-papillary level with left ventricular walls labeled.
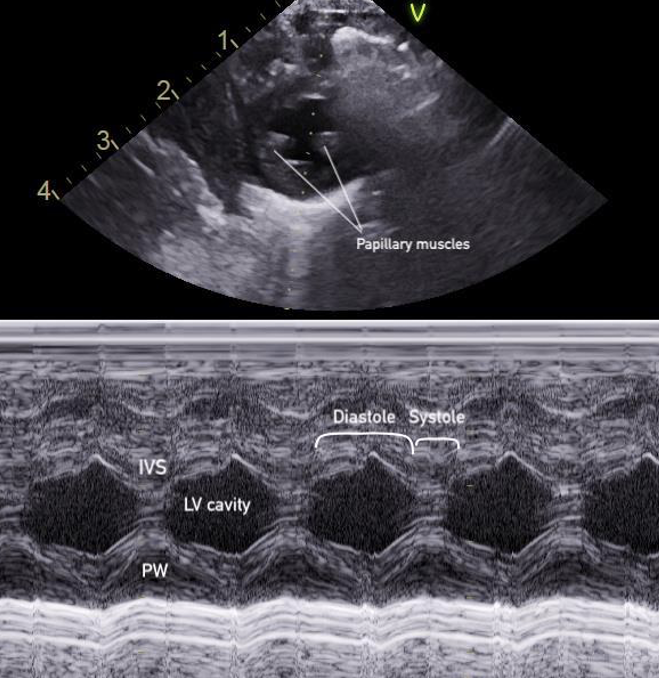 Figure 15: M-mode of the PSAX mid-papillary level
Figure 15: M-mode of the PSAX mid-papillary level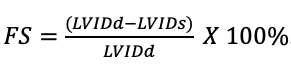
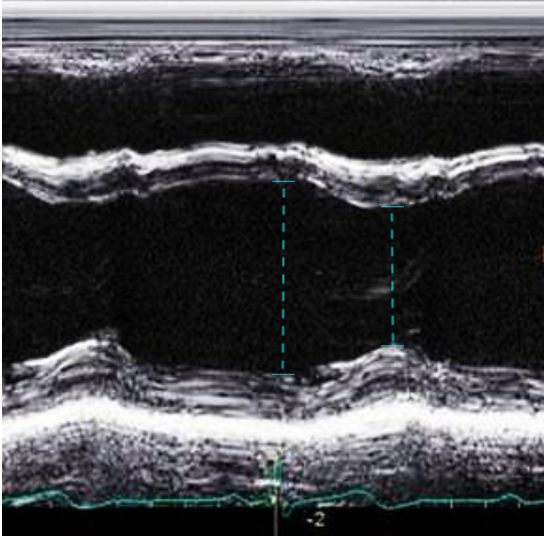 Figure 16. LVIDd and LVIDs caliper placement for FS/EF calculation in PSAX M-mode
Figure 16. LVIDd and LVIDs caliper placement for FS/EF calculation in PSAX M-mode

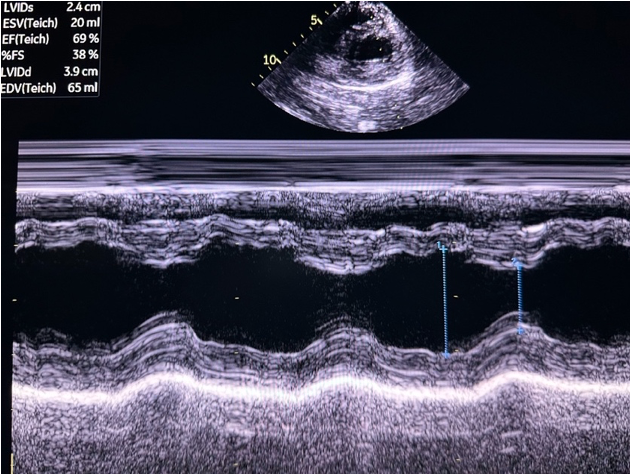 Figure 17. PSAX M-mode normal FS and EF
Figure 17. PSAX M-mode normal FS and EF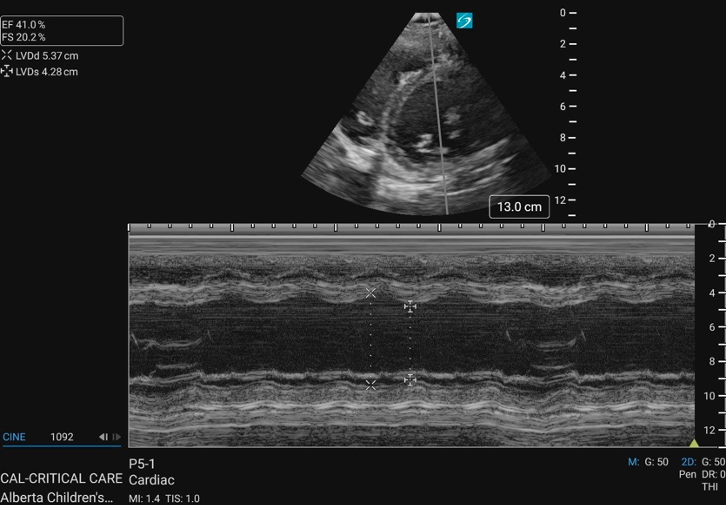 Figure 18. PSAX M-mode abnormal FS
Figure 18. PSAX M-mode abnormal FS
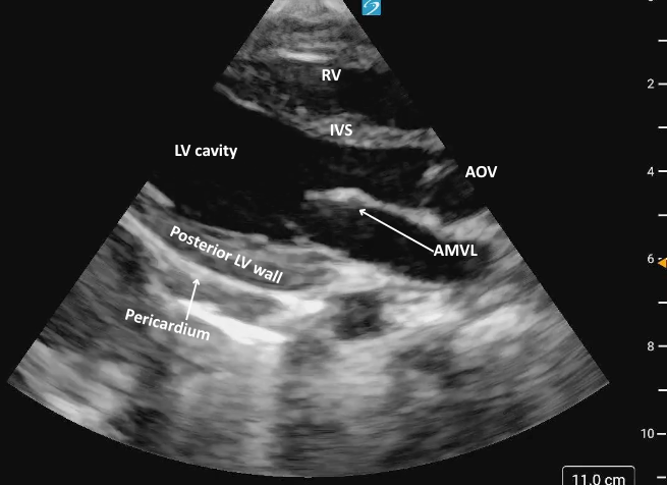 Figure 3: Parasternal long axis view in cardiology convention anatomically labeled.
Figure 3: Parasternal long axis view in cardiology convention anatomically labeled.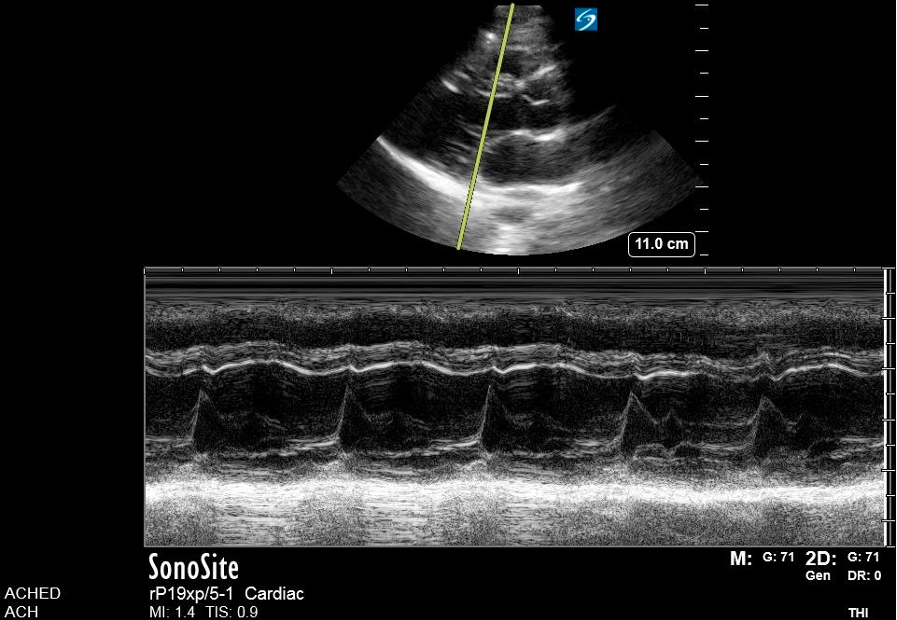 Figure 7. M-mode
Figure 7. M-mode 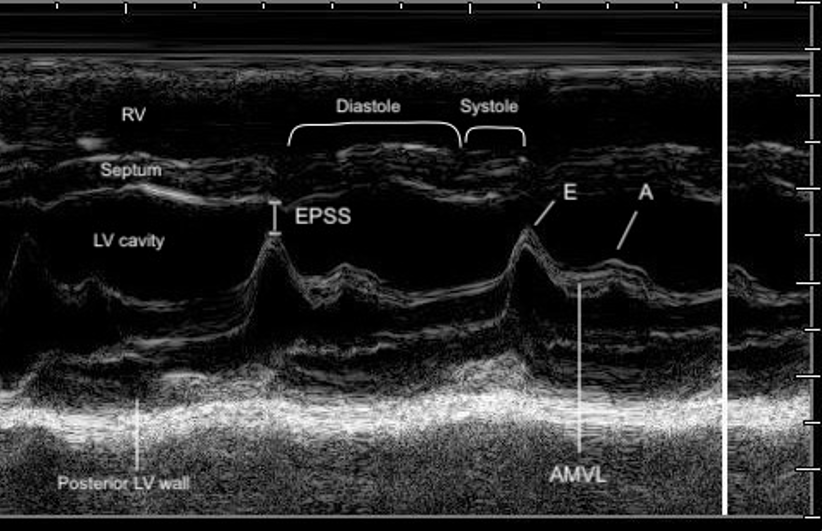 Figure 8. Zoomed-in view of the PLAX M-mode tracing at the level of the mitral valve tips.
Figure 8. Zoomed-in view of the PLAX M-mode tracing at the level of the mitral valve tips.
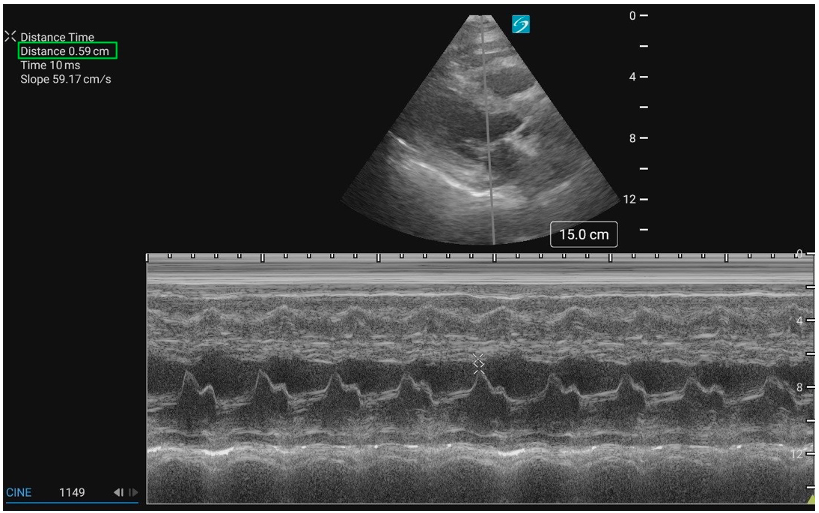 Figure 9: PLAX M-mode with normal EPSS measurement
Figure 9: PLAX M-mode with normal EPSS measurement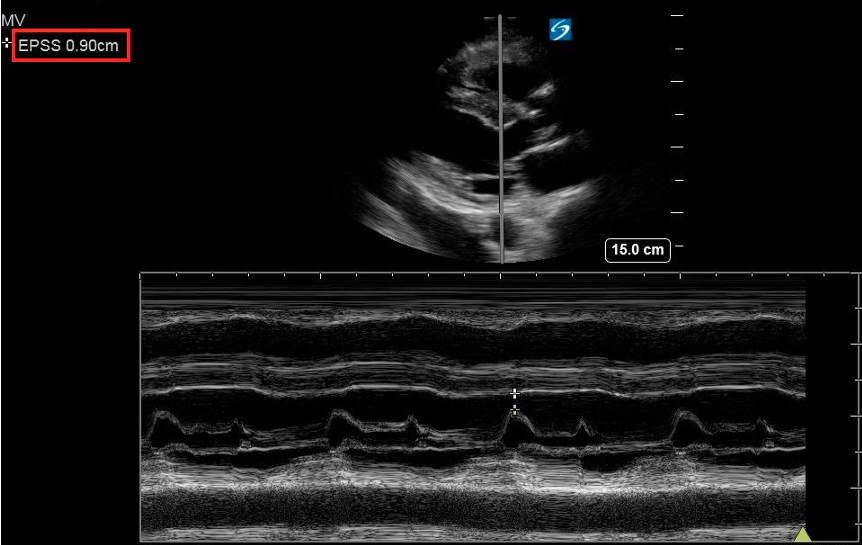 Figure 10: PLAX M-mode abnormal EPSS
Figure 10: PLAX M-mode abnormal EPSS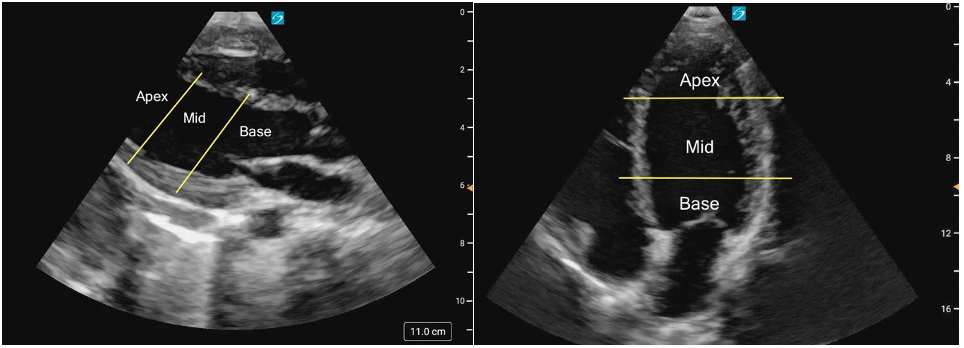 Figure 1ab: LV levels in the PLAX view (a) and A4C (b) in cardiology convention
Figure 1ab: LV levels in the PLAX view (a) and A4C (b) in cardiology convention